Effective magnetic moment (B.M. per Nickel) versus temperature for the... | Download Scientific Diagram

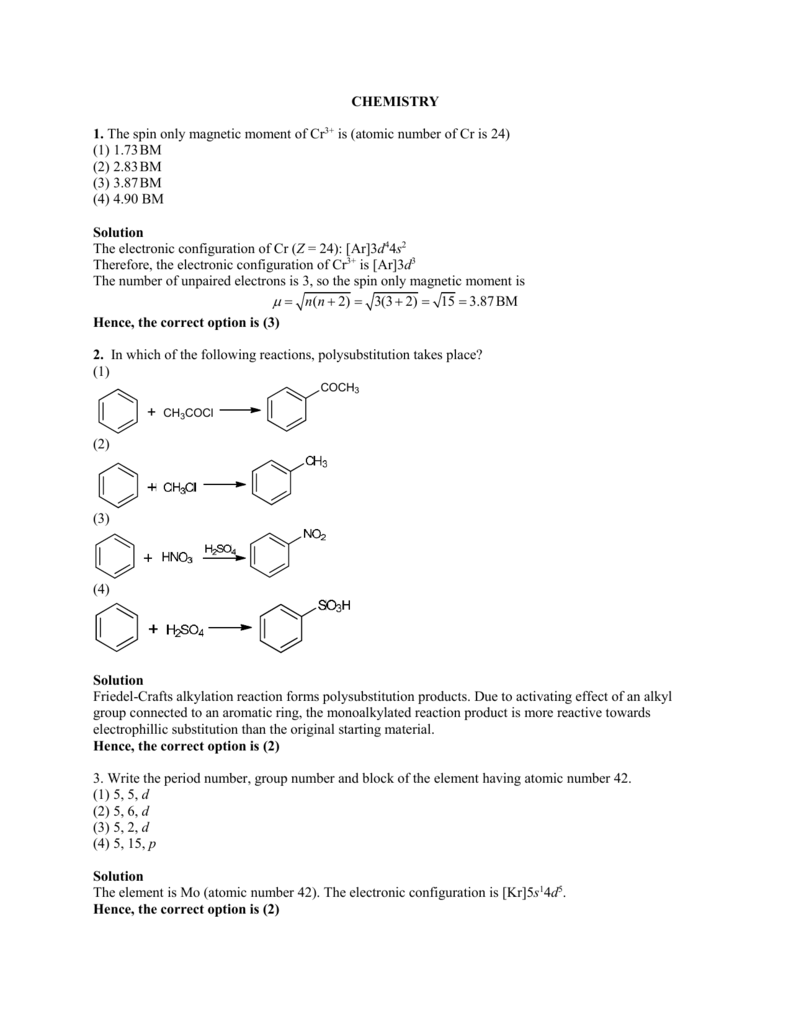
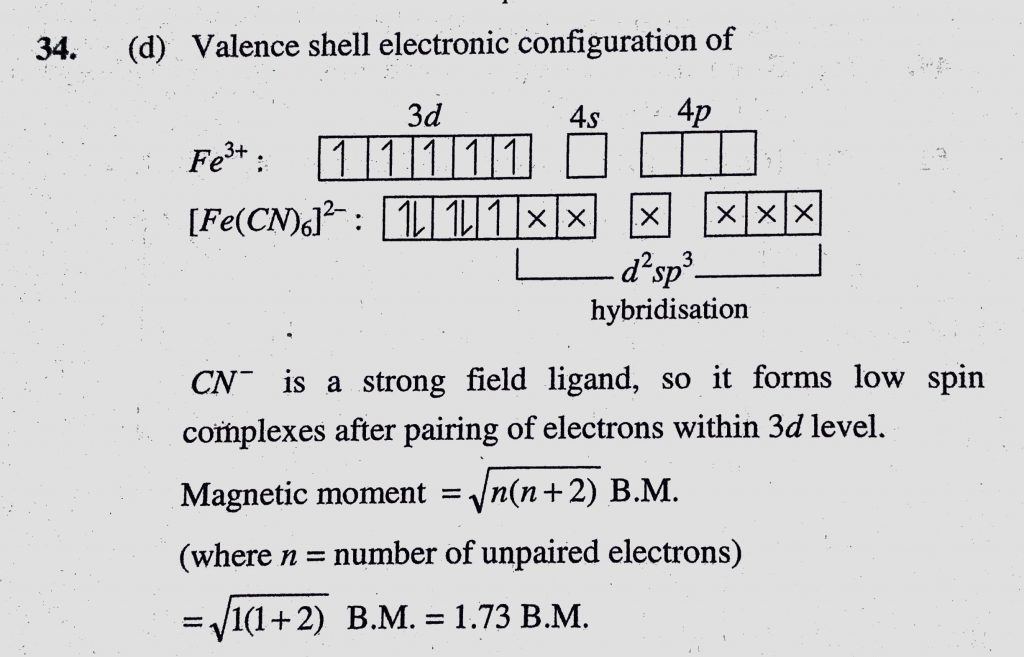
The magnetic moment of a transition metal ion is root 15 B.M. Therefore the number of unpaired electrons present in it is (a) 4 (b) 1 (c) 2 (d) 3 - Sahay Sir
27. Magnetic moment of an ion M^{3+} is 5.92BM. Numberof electrons in d-orbital of M will be(1) 4 (2) 5(3) 6 (4) 3 | Snapsolve
The complex showing a spin-only magnetic moment of 2.82 BM is - Sarthaks eConnect | Largest Online Education Community

Magnetic moment 2.83 BM is given by which of the following ions?(At. no.: Ti =22; Cr=24; Mn=25; Ni=28) - Wired Faculty

The correct electronic configuration and spin-only magnetic moment (BM) of Gd^3+ (Z = 64), respectively, are:
A compound of vanadium has a magnetic moment of 1.73 BM. Work out the electronic configuration of the vanadium ion in the compound. - Sarthaks eConnect | Largest Online Education Community
![SOLVED:Calculate the spin-only magnetic moment; /s (a.k.a. plp8), for the complex [Co(CNJ] Number 2.83 BM SOLVED:Calculate the spin-only magnetic moment; /s (a.k.a. plp8), for the complex [Co(CNJ] Number 2.83 BM](https://cdn.numerade.com/ask_images/b69b63ae151a47c3917e6e2d493e3bbb.jpg)
SOLVED:Calculate the spin-only magnetic moment; /s (a.k.a. plp8), for the complex [Co(CNJ] Number 2.83 BM

A compound of Magnease has a spin magnetic moment of 3.87 BM . if the manganese ion in the compound is - Brainly.in
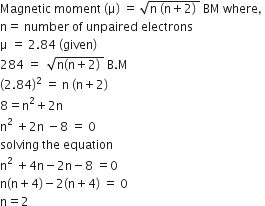
Magnetic moment 2.84 BM is given by (At. no. Ni= 28, Ti= 22, Cr=24, Co = 27) from Chemistry NEET Year 2015 Free Solved Previous Year Papers
![The spin only magnetic moment of [ZCl4]^2 - is 3.87 BM where Z is: The spin only magnetic moment of [ZCl4]^2 - is 3.87 BM where Z is:](https://d2rrqu68q7r435.cloudfront.net/images/3039131/570d1e82-cfd4-4271-bfe7-76de435db67c.jpg)



![SOLVED] The magnetic moment of Cu + is zero 173 BM 282 BM 387 B - Self Study 365 SOLVED] The magnetic moment of Cu + is zero 173 BM 282 BM 387 B - Self Study 365](https://www.extramarks.com/stryde/uploadfiles/Image/2009/03/18/2009031849001900123737139809020911.gif)


![Solved [NiCl2(PPh3)2] has a magnetic moment of 2.96 BM | Chegg.com Solved [NiCl2(PPh3)2] has a magnetic moment of 2.96 BM | Chegg.com](https://media.cheggcdn.com/media/6a5/6a5fdbdc-ebd2-4a71-a5d1-4113566c057f/phprXF3Ni)