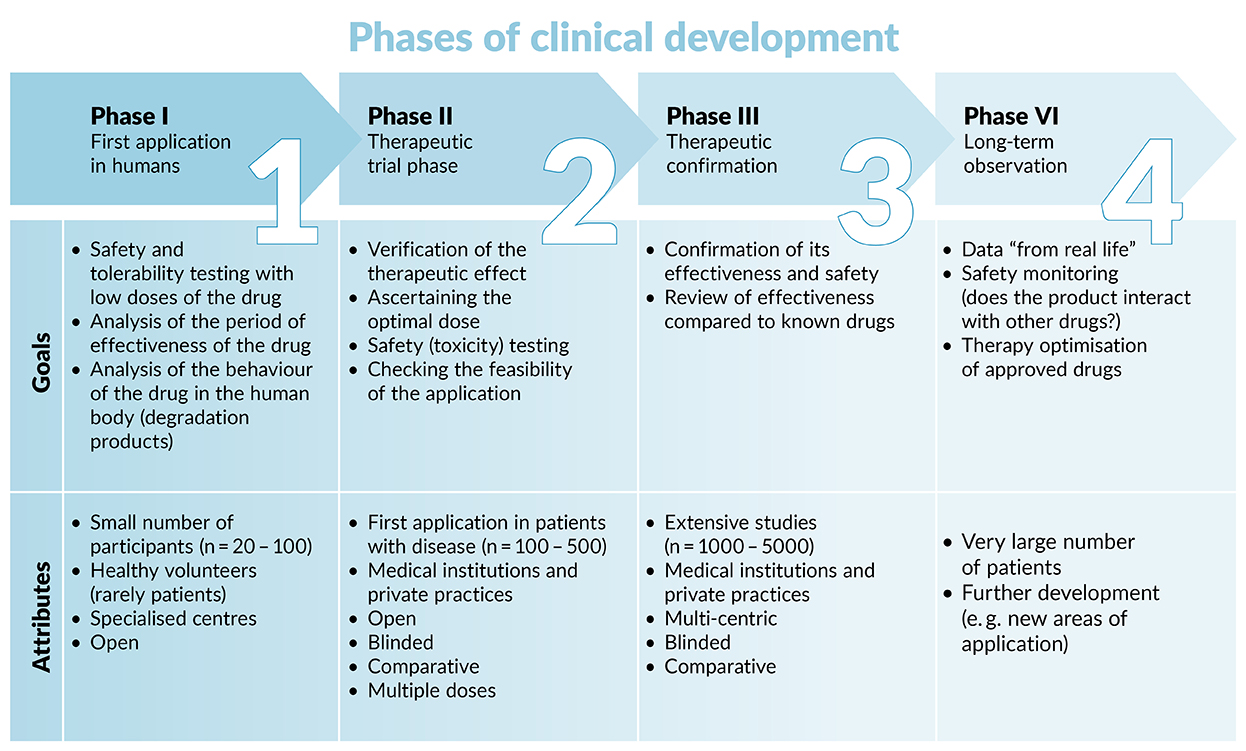
The International Society of Nephrology (ISN) - The ISN-ACT Clinical Trials Toolkit is an open-access, online resource to assist clinical research studies and trial site participation. Step-by-step guidance and best practices in: •

Study designs of registered clinical trials of products for SARS-CoV-2... | Download Scientific Diagram
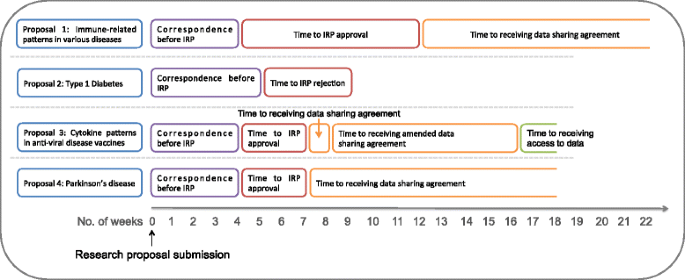
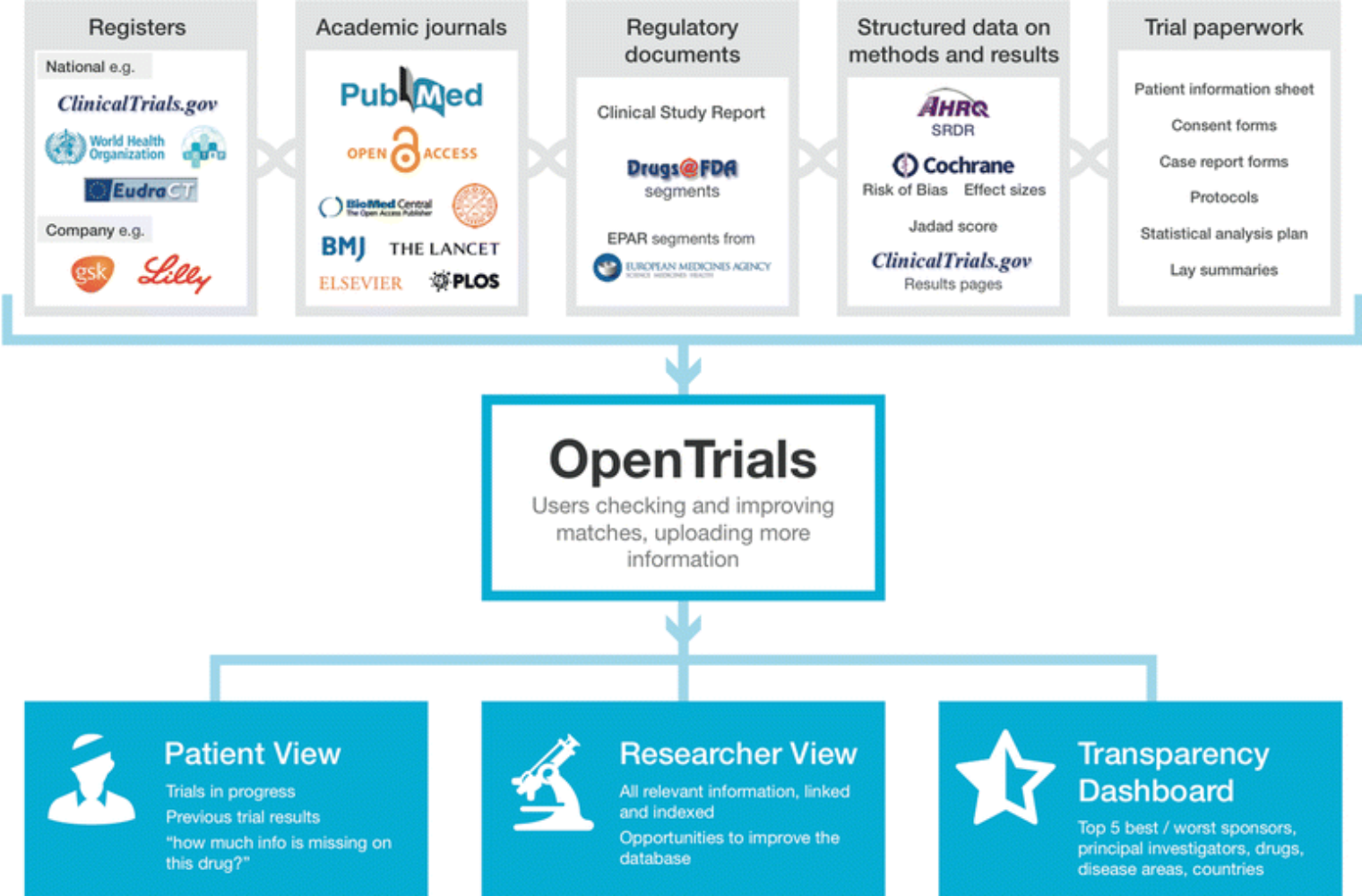
Opening clinical trial data: are the voluntary data-sharing portals enough? | BMC Medicine | Full Text

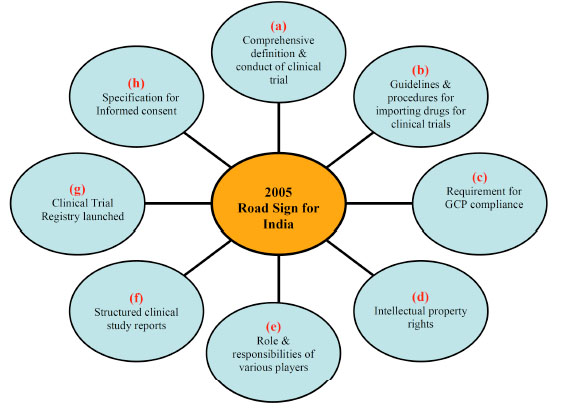
A critical appraisal of clinical trials conducted and subsequent drug approvals in India and South Africa | BMJ Open

Statistical controversies in clinical research: limitations of open-label studies assessing antiangiogenic therapies with regard to evaluation of vascular adverse drug events—a meta-analysis - Annals of Oncology

Schematic view of an open label extension to a randomized controlled... | Download Scientific Diagram

Efficacy and safety of hydroxychloroquine as pre-and post-exposure prophylaxis and treatment of COVID-19: A systematic review and meta-analysis of blinded, placebo-controlled, randomized clinical trials. - The Lancet Regional Health – Americas